Update
Published in spanish Científica Dental Vol. 20. Nº 2. 2023
www.cientificadental.es
Antiplatelet therapy and oral surgery: to discontinue or not, that is the question
SUMMARY
Cardiovascular diseases are one of the most prevalent systemic disorders in the Western world. Many cardiopathy patients have had an acute coronary episode and are being treated with platelet antiaggregants. The therapy with these drugs can be a challenge for the odontologist, who must face an important dilemma: either maintain the drug with the consequent haemorrhagic risk, or withdraw it, with the possibility of thromboembolic complications, posing a risk to the patient’s life. Therefore, odontologists should know what should be the management of this type of patients before the prospect of performing a surgical procedure in the oral cavity or even a simple tooth extraction.
The objectives of this narrative review are, firstly, to recall the platelet physiology and the platelet thrombus formation mechanisms; secondly, to delve into the action mechanisms of the different antiplatelet drugs; and, thirdly, since there are no clinical guidelines on the subject, make a critical approach to the existing guidelines for a dental management of this type of patients, in order to prevent the occurrence of possible complications, not only local, but, more importantly, systemic complications. In these cases, before withdrawing antiplatelet therapy, it would be advisable to reduce the haemorrhagic risk versus the risk of generating a new thromboembolic episode, such as stent thrombosis or recurrence of the acute coronary accident, events that could put the patient’s life at risk.
Cardiovascular disease is the systemic disorder that generates greater morbidity and mortality in the western world. In Spain, according to data from the National Institute of Statistics, in 2020, cardiovascular disease was the leading cause of death among the general population, above tumours and COVID-19. In this sense, ischemic heart disease is the leading cause of death in men and stroke in women.
Among cardiovascular diseases, atherosclerotic pathology is the main cause of morbimortality in our country, including acute coronary syndromes, cerebrovascular diseases and peripheral arterial disease1. The etiopathogenic mechanism underlying all these pathologies is the rupture of the atheroma plaque, which triggers the platelet aggregation which is the cause of the acute thrombosis process. For this reason, in recent years, new antiplatelet drugs have been developed, which constitute the cornerstone of the recurrence prevention of acute ischemic episodes, both short-term and long-term. These drugs have to be known by the odontologist, especially when performing a surgical procedure, to know what should be the correct dental management to avoid potential complications, not only from the point of view of local bleeding, but also, most importantly, to avoid systemic complications (such as stent thrombosis or the appearance of a new thromboembolic event), which could put the patient’s life at risk2.
The purpose of this narrative review is, firstly, to recall the platelet physiology and the platelet thrombus formation mechanisms; secondly, to delve into the mechanisms of action of the different platelet antiaggregants; and, thirdly, since there are no clinical guidelines in this regard, make a critical approach to the existing guidelines for their correct dental management, in order to perform a surgical procedure in the oral cavity or even a simple tooth extraction, with sufficient guarantees of success.
Physiology of the platelet
The platelet is one of the blood forming elements, along with red and white blood cells. Normally, there are between 150,000 and 400,000 platelets per mcL in the blood, and the average platelet volume is usually 7-9 cubic micrometers. Platelets come from the hematopoietic stem cells of the bone marrow, specifically from the myeloid lineage and have an average life of 7 to 10 days. Inside they have alpha granules and dense granules, where molecules of special relevance accumulate in platelet physiology3.
Platelets play a primary role in haemostasis, since they initiate the repair of vascular lesions, forming the platelet plug, and also promote blood clotting, through the activation of thrombin released from the platelets themselves and the calcium released from the dense granules which are necessary for the formation of fibrin3.
Platelet physiology involves several enzymes, such as cyclooxygenase (COX), which transforms arachidonic acid (AA) from membrane phospholipids, in prostaglandins (PG), which are the thromboxane A2 (TXA2), vasoconstrictor and platelet proaggregant and prostacyclin (PGI2), which is a vasodilator and antiaggregant and originates in the vascular endothelium3. Other platelet enzymes include: phospholipase A2, which releases AA from membrane phospholipids and the phosphodiesterase, which hydrolyzes the cAMP.
The platelets have receptors in their membrane which are glycoproteins (GP) and are inactive under normal conditions. The most relevant are GP Ia and GP VI that bind to collagen, GPIb that binds to the von Willebrand factor (VWF) and GP IIb/IIla, which binds to several proteins, but the most important is fibrinogen. These receptors are involved in platelet adhesion phenomena (platelet binding to the injured vessel), activation (change of platelet morphology causing the secretion of granules) and aggregation (binding between several platelets)3.
Platelet thrombus formation mechanism
Platelets circulate in the bloodstream inactively. But when a vessel injury occurs, subendothelial collagen is exposed, which is the stimulus to recruit the platelets that will form the platelet plug. Remember that platelets do not adhere to the intact endothelium, but they can adhere to a foreign body inside the bloodstream (such as a coronary stent or a prosthetic heart valve)3.
Adhesion: When a vessel is injured, the circulating platelets slow down their speed over the damaged area, against the blood flow that pushes them, thanks to the platelet GPIb binding to the von Willebrand factor (VWF) of the matrix under the endothelium. Then, the subendothelial collagen establishes a more stable binding by binding to platelets3. GPIb and GPVI.
Activation: After adhesion, platelet activation occurs, appearing on the exterior of the receptors that were inactive. These activate intracellular molecules, which cause a change in platelet morphology, with the emission of pseudopods and release of certain substances that promote platelet aggregation, perpetuating the process. These molecules, known as platelet agonists are: TXA2, ADP and thrombin. Of all of them, ADP is the most potent for recruiting platelets and spreading arterial thrombus, for which it is considered a platelet activation amplifier. Platelets have on their surface three receptors for ADP: P2Y1, P2Y12 and P2X. Each induces different platelet signalling pathways, but P2Y12 is the most important since it favours the release of the content of the granules, the increase of intracellular calcium, the generation of TXA2 and the activation of the GPIIb-IIIa receptor, which is key in platelet aggregation. Consequently, the platelet P2Y12 receptor blockage is crucial to inhibit platelet activation and aggregation and thus prevent platelet thrombus formation. Therefore, in recent years new drugs have been developed capable of blocking this receptor1.
Release: After activation, the molecules stored in the granules of the platelets are released. Activated platelets can release up to 300 different proteins. From alpha granules, proteins homologous to plasma (fibrinogen, fibronectin, factor XIII, VWF) and platelet-specific proteins (platelet factor 4-FP4), thromboglobulin, P-selectin, PDGF (platelet-derived growth factor) and thrombospondin) are released. From the dense granules ADP, ATP, calcium and serotonin (5-hydroxytryptamine or 5-HT) is released3.
Aggregation: Once the platelets are trapped in the damaged area, new platelets are recruited from the bloodstream, known as platelet aggregation. Activation of the GPIIb/IIIa receptor is the final pathway leading to platelet aggregation. Once activated, it binds to its ligands, which have the sequence of RGD amino acids (Arg-Gly-Asp or arginine-glycine-aspartic), such as fibrinogen, but also VWF, fibronectin and vitronectin. This receptor is specific to platelets and binds in a bivalent manner to fibrinogen, forming binding bridges between two platelets3.
Regarding the mechanisms that regulate platelet aggregation, there are the contribution and inhibition factors.
Thus, they favour the platelet aggregation the ADP, the thrombin, the collagen, adrenaline and TXA2. While cAMP, cGMP and PGI2 inhibit it, as well as nitric oxide (NO), which in addition to being a platelet antiaggregant, is considered the most potent vasodilator of the organism3.
Platelet antiaggregants drugs (APAs)
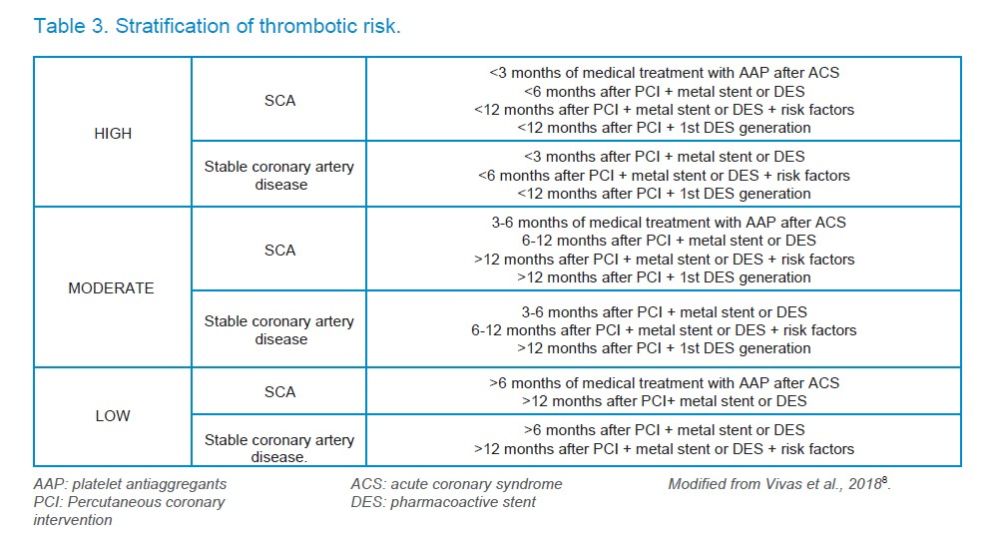
Antiplatelet agents (APAs) are drugs that inhibit platelet aggregation, acting as antithrombotics. They usually act irreversibly and their function cannot be monitored, but it is usually considered that their effect lasts as long as the average life of the platelet, that is, between 7 and 10 days1. The main indications of these drugs include acute coronary syndromes (ACS) (which include acute myocardial infarction-AMI and unstable angina), stable coronary artery disease, cerebrovascular disease, and peripheral artery disease, but they are also used after surgical treatments that are performed after the appearance of these conditions, such as percutaneous coronary intervention (PCI) or revascularization surgery, and in the prevention of recurrence of the same, that is, in the secondary prophylaxis of atherosclerotic disease1.
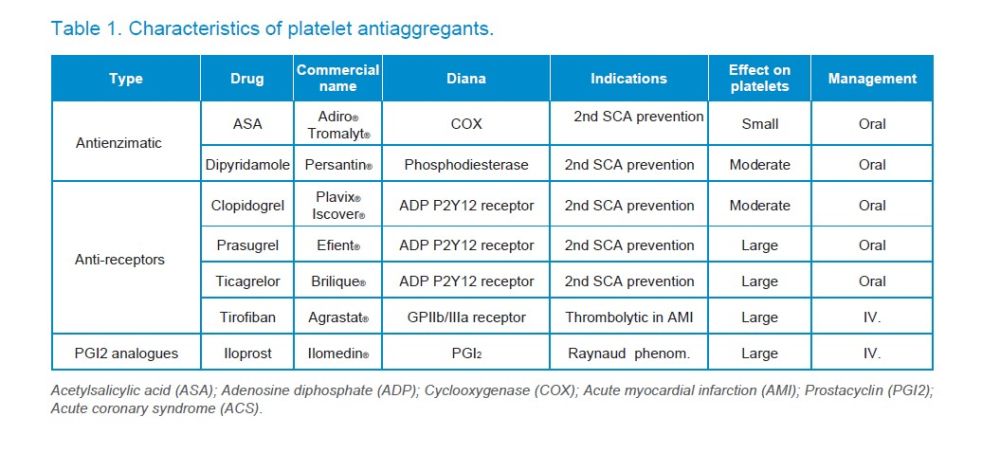
AAPs are drugs whose action mechanism is based on inhibiting platelet enzymes, such as COX (acetylsalicylic acid) or phosphodiesterase (dipyridamole), on the platelet P2Y12 receptor (such as thienopyridines) or GPIIb/IIIa receptor (such as tirofiban) or in acting as analogues of molecules that inhibit platelet aggregation (such as iloprost) (Table 1).
APAs can be classified according to their action mechanism in4-7:
1. Antienzimatic
1.1. Cyclooxygenase (COX) inhibitors
1.1.1. Acetylsalicylic acid (ASA) (Aspirin®, Adiro®, Tromalyt®)
1.1.2. Triflusal (Disgren®)
1.2. Phosphodiesterase inhibitors
1.2.1. Dipyridamole (Persantin®)
1.2.2. Cilostazol
1.2.3. Pentoxifylline
2. Receptor inhibitors
2.1. Of ADP (P2Y12): Ticlopidine, Clopidogrel, Prasugrel, Ticagrelor
2.2. From GPIIb/IIIa: Abciximab, Tirofiban, Eptifibatide
2.3. Of thrombin (PAR1): Vorapaxar
3. Prostacyclin analogues
3.1. Iloprost (Ilomedin®)
-
- Antienzimatic
1.1. COX inhibitors
1.1.1. Acetylsalicylic acid (Adiro® and Tromalyt®) Acetylsalicylic acid (ASA) is the antiplatelet par excellence. It is an irreversible inhibitor of the cyclooxygenase COX-1 and COX-2 of the platelet and, therefore, inhibits the synthesis of TXA2 platelet and of the PGI2 of the vascular endothelium, but especially of the first.
- Antienzimatic
In addition, small doses appear to affect only TXA2. Also, since platelets do not have a nucleus, they do not have the ability to resynthesize COX, unlike endothelial cells, for which the TXA2 inhibition lasts for the average platelet life, that is, between 7 and 10 days.
Inhibition of TXA2 only suppresses one of the aggregation mechanisms, but does not affect the aggregation induced by ADP. However, another effect of ASA in the
platelets is that it decreases the secretion of dense granules, that is, decreases the release of proaggregant substances during platelet activation. This would explain why its effects on platelets are more than one would expect from simple platelet inhibition dependent on a relatively weak agonist such as TXA21.
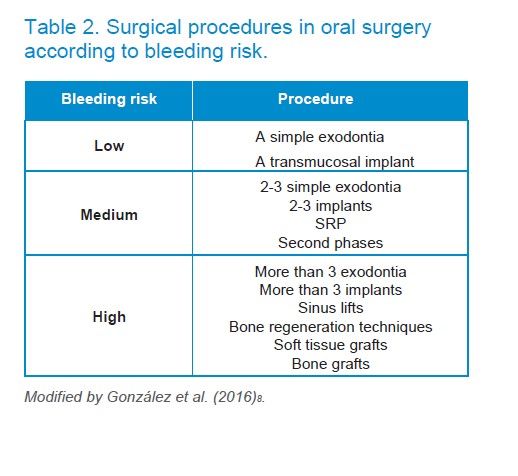
ASA is the basic antithrombotic therapy, used as an antiplatelet treatment only in the secondary prevention of atherosclerotic disease8.
1.1.2. Triflusal (Disgren®)
Triflusal is an ASA analogue, which selectively inhibits the platelet COX, but does not affect endothelial cells. Triflusal has fewer side effects than ASA, so it is indicated in patients with ASA resistance and in geriatric patients.
1.2. Phosphodiesterase inhibitors
1.2.1. Dipyridamole (Persantin®)
Dipyridamole is a phosphodiesterase inhibitor, which increases intracellular cAMP levels, inhibiting aggregation; it is also a vasodilator. It does not have advantages over ASA, but it can be associated with anticoagulant drugs and given to patients with cardiac valve prostheses with ASA intolerance.
1.2.2. Cilostazol
It increases intracellular cAMP levels and is a vasodilator.
1.2.3. Pentoxifylline
Pentoxifylline is a vasodilator phosphodiesterase inhibitor, currently used in the prevention of jaw osteonecrosis.
2. Platelet receptor inhibitors
2.1. Inhibitors of ADP P2Y12
2.1.1. Irreversible inhibitors: Thienopyridines
2.1.1.1. 1st Generation: Ticlopidine (Tiklid®) Ticlopidine is a thienopyridine derivative, which behaves like a prodrug, that is, it is metabolised in the liver resulting in an active
metabolite, which antagonizes ADP induced aggregation. It was the first inhibitor of the P2Y12 receptor, but the relative frequency of adverse reactions, such as diarrhoea and, above all, neutropenia (in 0.8% of cases), has made its use increasingly low.
2.1.1.2. Of 2nd generation: Clopidogrel (Plavix®, Iscover®)
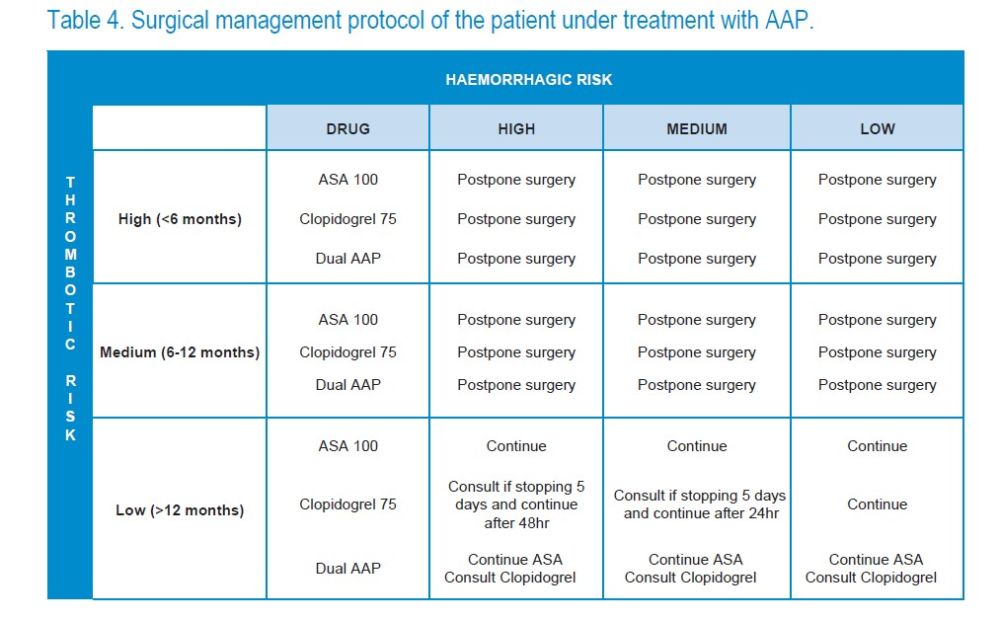
It is a prodrug, which requires two oxidation reactions in the liver to transform into the active metabolite, which inhibits the P2Y12 receptor. However, a large individual variability has been described in the induced antiaggregation response by clopidogrel. It is usually used at a dose of 75 mg a day, being more powerful than 100 mg of ASA.
A coronary stent may be administered in conjunction with the ASA for the treatment of ACS after placing or after percutaneous revascularization surgery, which constitutes the so-called “dual antiplatelet therapy” or “dual platelet antiaggregation” (DPA).
2.1.1.3. Of 3rd generation: Prasugrel (Efient®)
Prasugrel is another prodrug that inhibits the P2Y12 receptor, and is more potent, faster and has less variability in the antiplatelet response than clopidogrel. It is the only one that has benefits in diabetics.
2.1.2. Reversible inhibitors
2.1.2.1. Ticagrelor (Brilique®)
Ticagrelor is an antagonist of the P2Y12 receptor with a reversible effect. It is faster and more powerful than clopidogrel. In addition, it has extraplatelet effects that are beneficial from a cardiovascular point of view.
2.1.2.2. Cangrelor
Recently, new antagonists even more potent antagonists like Cangrelor and Elinogrel have been designed. These new antiaggregants achieve greater antithrombotic efficacy, but also involve an increased risk of bleeding.
2.2. GPIIb/IIIa receptor inhibitor
2.2.1. Abciximab, Tirofiban, Eptifibatide
They are antiaggregants for hospital use administered by IV, blocking the binding of fibrinogen and VWF to the glycoproteins of the platelet surface (mediated by the GPIIb/IIIa receptor).
They are used as emergency thrombolytic drugs in the treatment of AMI. The sooner therapy with GPIIb/IIIa inhibitors is provided, the most favourable the prognosis for AMI.
2.3. PAR1 antagonist (protease 1 activating receptor)
2.3.1. Vorapaxar (Zontivity®)
It inhibits thrombin-mediated aggregation, since it is an antagonist of the thrombin PAR1 receptor. Accepted by the FDA, but not by the EMA.
3. Prostacyclin analogues
3.1. Iloprost (Ilomedin®)
Iloprost is an analogue of prostacyclin, which increases intraplatelet cAMP and is also a vasodilator. It is used in peripheral artery disease, thromboangiitis obliterans and Raynaud’s disease.
Badimon L, Vilahur G. Mecanismos de acción de los diferentes agentes antiplaquetarios (Action mechanisms of different antiplatelet agents). Rev Esp Cardiol 2013;13(B):8-15.
Ferrari E, Benhamou M, Cerboni P, Marcel B. Coronary syndromes following aspirin withdrawal: a special risk for late stent thrombosis. J Am Coll Cardiol 2005; 45: 456- 9.
López Farré A, Macaya C. Plaqueta: Fisiología de la activación y la inhibición. Rev Esp Cardiol 2013;13(B):2-7.
Jourdi G, Godier A, Lordkipanidze M, Marquis-Gravel G, Gaussem P. Antiplatelet Therapy for Atherothrombotic Disease in 2022. From Population to Patient-Centered Approaches. Front Cardiovasc Med. 2022 https://doi. org/10.3389/fcvm.2022.805525.
Badimon L, Mendieta G, Vilahur G. Diferencias en los mecanismos de acción de los nuevos antiagregantes: ¿cómo actúan? Rev Esp Cardiol Supl 2014;14 (A):3-9.
Ganthous AE, Ferneini EM. Aspirin, Plavix, and Other Antiplatelet Medications. What the Oral and Maxillofacial Surgeon Needs to Know. Oral Maxillofacial Surg Clin N Am. 2016 http://dx.doi.org/10.1016/j. coms.2016.06.003
Mahmood H, Siddique I, McKechnie A. Antiplatelet drugs: A review of pharmacology and the perioperative management of patients in oral and maxillofacial surgery. Ann R Coll Surg Engl 2020; 102(1): 9–13.
Vivas D, Roldán I, Ferrandis R, et al. Manejo perioperatorio y periprocedimiento del tratamiento antitrombótico: documento de consenso de SEC, SEDAR, SEACV, SECTCV, AEC, SECPRE, SEPD, SEGO, SEHH, SETH, SEMERGEN, SEMFYC, SEMG, SEMICYUC, SEMI, SEMES, SEPAR, SENEC, SEO, SEPA, SERVEI, SECOT
y AEU. Rev Esp Cardiol. 2018;71(7):553- 564.
González Fernández-Tresguerres F, Alvarez Sirvent A, Torres J, Fernández- Tresguerres I. Nuevos anticoagulantes orales: repercusión odontológica. Científica Dental 2016;13:35-43.
Olmos-Carrasco O, Pastor-Ramos V, Espinilla-Branco R, et al. Hemorrhagic complications of dental extractions in 181 patients undergoing double antiplatelet therapy. J Oral Maxillofac Surg 2015;73:203-10.
Cardona-Tortajada F, Sainz-Gomez E, Figuerido-Garmendia J, et al. Dental extractions in patients on antiplatelet therapy. A study conducted by the Oral Health Department of the Navarre Health Service (Spain). Med Oral Patol Oral Cir Bucal 2009;14:e588.
Sanchez-Palomino P, Sanchez-Cobo P, Rodrigues-Archilla A, et al. Dental extraction in patients receiving dual antiplatelet therapy. Med Oral Patol Oral Cir Bucal 2015;20:e616.
Yanamoto S, Hasegawa T, Rokutanda S, et al. Multicenter Retrospective Study of the Risk Factors of Hemorrhage After Tooth Extraction in Patients Receiving Antiplatelet Therapy. J Oral Maxillofac Surg 2017;75:1338-43.
Iakovou I, Schmidt T, Bonizzoni E et al. Incidence, predictors, and outcome of thrombosis after successful implantation of drug-eluting stents. JAMA 2005;293(2):126–130.
Omar HR, Socias SM, Powless, RA, Sprenker C, Karlnoski R, Mangar D, Camporesi EM. Clopidogrel is not associated with increased bleeding complications after full-mouth extraction: A retrospective study. J Am Dent Assoc. 2015; 146:303-9.
Lu SY, Tsai CY, Lin LH, Lu SN. Dental extraction without stopping single or dual antiplatelet therapy: Results of a retrospective cohort study. Int J Oral Maxillofac Surg. 2016;45:1293-8.
Eapen BV, Baigi MF, Avinash S. An Assessment of the Incidence of Prolonged Postoperative Bleeding After Dental Extraction Among Patients on Uninterrupted Low Dose Aspirin Therapy and to Evaluate the Need to Stop Such Medication Prior to Dental Extractions. J Maxillofac Oral Surg. 2017;16(1):48-52.
Gupta R, Dugal A, Sane VD, Hiwarkar S, Khandelwal S, Iyengar A. Effect of Low-Dose Aspirin on Bleeding Following Exodontia: A Prospective Clinical Study. J Maxillofac Oral Surg. 2018;17(3):350- 355.
Burger W, Chemnitius M, Kneissl GD, Rucker G. Low-dose aspirin for secondary cardiovascular prevention- cardiovascular risks after its perioperative withdrawal versus bleeding risks with its continuation- review and meta-analysis. J Int Med. 2005;257:399-414.
Biondi-Zoccai GG, Lotrionte M, Agostoni P, et al. A systematic review and meta-analysis on the hazards of discontinuing or not adhering to aspirin among 50,279 patients at risk for coronary artery disease. Eur Heart J 2006;27(22):2667–74.
Pennacchi M, Stio RE, Lucisano L, Calcagno S, Mancone M, Sardella G. Five years of dual antiplatelet therapy DES thrombosis after clopidogrel withdrawal. Int Heart J 2013;54:234-6.
Artang R, Dieter RS. Analysis of 36 reported cases of late thrombosis in drug-eluting stents placed in coronary arteries. Am J Cardiol 2007; 99(8):1039–43.
Lillis T, Ziakas A, Koskinas K, Tsirlis A, Giannoglou G. Safety of dental extractions during uninterrupted single or dual antiplatelet treatment. Am J Cardiol 2011; 108:964-7.
Napeñas JJ, Oost FC, DeGroot A, et al. Review of postoperative bleeding risk in dental patients on antiplatelet therapy. Oral Surg Oral Med Oral Pathol Oral Radiol 2013; 115:491–9.
Nathwani S, Martin K. Exodontia in dual antiplatelet therapy: the evidence. Br Dent J 2016;220(5):235-8.
Ockerman A, Bornstein MM, Leung YY, Li SKY, Politis C, Jacobs R. Incidence of bleeding after minor oral surgery in patients on dual antiplatelet therapy: a systematic review and meta- analysis. Int J Oral Maxillofac Surg 2020;49(1):90-8.
Grines CL, Bonow RO, Casey DE Jr., et al. Prevention of premature discontinuation of dual antiplatelet therapy in patients with coronary artery stents: a science advisory from the American Heart Association, American College of Cardiology, Society for Cardiovascular Angiography and Interventions, American College of Surgeons, and American Dental Association, with representation from the American College of Physicians. J Am Dent Assoc 2007;138(5):652-5.
Childers CP, Maggard-Gibbons M, Ulloa JG et al. Perioperative management of antiplatelet therapy in patients undergoing non-cardiac surgery following coronary stent placement: a systematic review. Syst Rev 2018;7:4.
Park M, Her S, Kwon J. Safety of dental extractions in coronary drug-eluting stenting patients without stopping multiple antiplatelet agents. Clin Cardiol 2012; 35:225-230.
Sáez-Alcaide LM, Sola C, Molinero- Mourelle P, Paredes-Rodríguez V, Zarrias-Caballero C, Hernández- Vallejo G. Dental management in patients with antiplatelet therapy: A systematic review. J Clin Exp Dent 2017;9(8):e1044-e1050.
Wahl MJ. Dental surgery and antiplatelet agents: bleed or die. Am J Med 2014; 127:260-7.